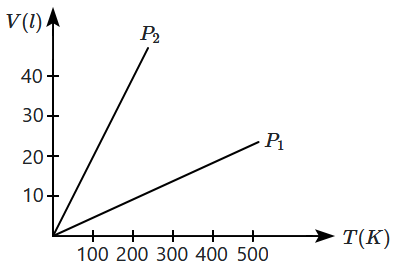
The volume versus temperature graphs for a given mass of an ideal gas are shown in the figure at two different values of constant pressure. What can be inferred about relation between \(\mathrm{P_1}\) and \(\mathrm{P_2}\)?
1. \(\mathrm{P_1}>\mathrm{P_2} \)
2. \(\mathrm{P_1}=\mathrm{P_2} \)
3. \(\mathrm{P_1}<\mathrm{P_2} \)
4. data is insufficient

© 2026 GoodEd Technologies Pvt. Ltd.