6.23 The choice of a reducing agent in a particular case depends on thermodynamics factor. How far do you agree with this statement? Support your opinion with two examples.
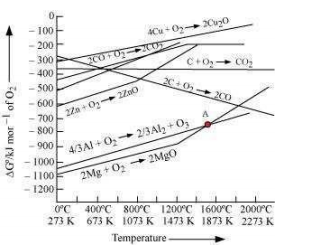 The above figure is a plot of Gibbs energy vs. T for formation of some oxides. It can be observed from the above graph that metal can reduce the oxide of other metals, if the standard free energy of formation of the oxide of the former is more negative than the latter. For example, since is more negative than , Al can reduce Cu2O to Cu, but Cu cannot reduce . Similarly, Mg can reduce ZnO to Zn, but Zn cannot reduce MgO because is more negative than
The above figure is a plot of Gibbs energy vs. T for formation of some oxides. It can be observed from the above graph that metal can reduce the oxide of other metals, if the standard free energy of formation of the oxide of the former is more negative than the latter. For example, since is more negative than , Al can reduce Cu2O to Cu, but Cu cannot reduce . Similarly, Mg can reduce ZnO to Zn, but Zn cannot reduce MgO because is more negative than 
© 2026 GoodEd Technologies Pvt. Ltd.