11.33 When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
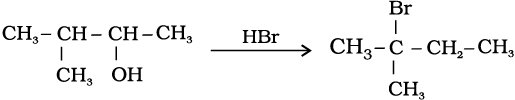
Give a mechanism for this reaction.
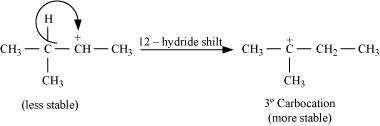
(Hint : The secondary carbocation formed in step II rearranges to a more stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.
The mechanism of the given reaction involves the following steps:
Step 1: Protonation
3- MethylbutAN-2-OF
Step 2: Formation of 2 carbocation by the elimentation of a water molecule
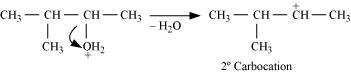
Step 3: Re-arrangement by the hydride-ion shift
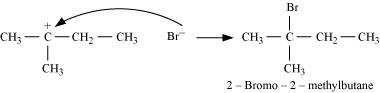
Step 4: Nucleophilic attack

© 2026 GoodEd Technologies Pvt. Ltd.